Corneal collagen crosslinking (CXL)
What is crosslinking?
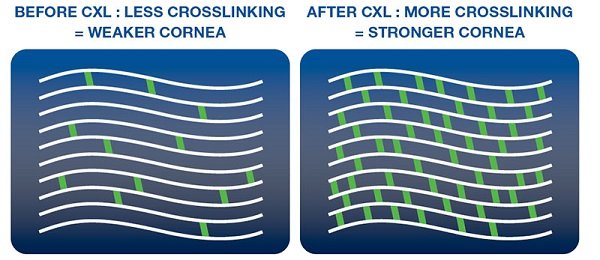
Corneal collagen crosslinking (CXL) is a minimally invasive treatment designed to strengthen a cornea weakened by disease. The primary use is for progressive keratoconus. Before the development of CXL, many patients with keratoconus had no option but to face ongoing progression and, in many cases, eventual corneal transplantation.
How long has CXL been used to treat Keratoconus?
Corneal crosslinking was first performed in 1998 after extensive research by Professor Theo Seiler at the University of Dresden in Germany. A clinical study followed between 1999 and 2003. In December 2005, Peschke, a Swiss company, introduced the first commercially available CXL device. While crosslinking research expanded across Europe and other countries soon after, the procedure did not receive FDA approval in the United States until April 2016.

What is Keratoconus?
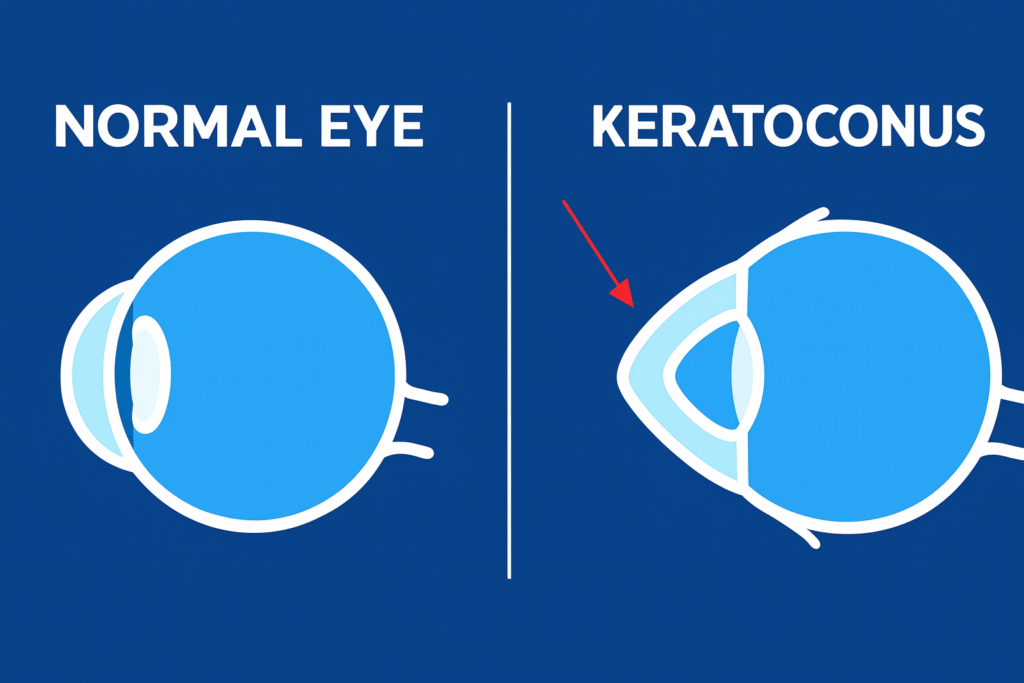
Keratoconus is a non-inflammatory condition that causes thinning of the cornea in both eyes, though often unevenly. It usually begins in the second decade of life and is marked by progressive corneal steepening, thinning, and scarring at the apex. As the disease advances, the cornea gradually assumes a cone-like shape.
Is Crosslinking the Standard of Care for Keratoconus?
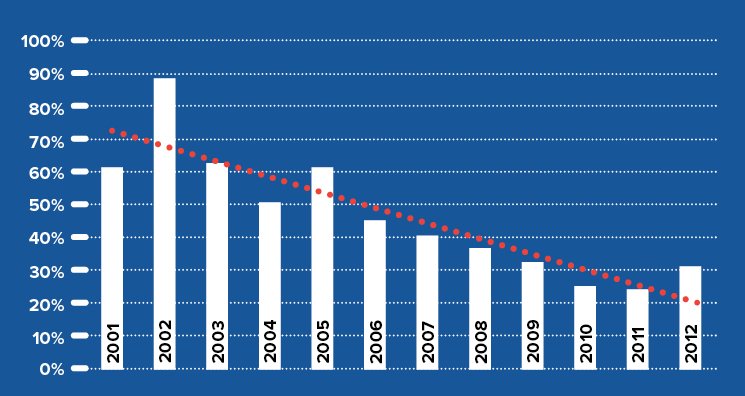
Traditionally, keratoconus in the U.S. was managed with hard contact lenses until progression led to intolerance or vision loss from scarring. This approach is now outdated. With the advent of corneal crosslinking (CXL), disease progression can be halted much earlier, and since 2005 CXL has become the global standard of care for progressive keratoconus. As a result, the need for full-thickness corneal transplantation has declined significantly in Europe.
Who is a candidate for crosslinking?
CXL is approved for progressive keratoconus. Because patients aged 10–20 are at highest risk of progression, early treatment is recommended to preserve long-term vision. After age 40, the cornea naturally stabilizes and CXL is rarely needed, though exceptions exist.
How is the procedure performed?
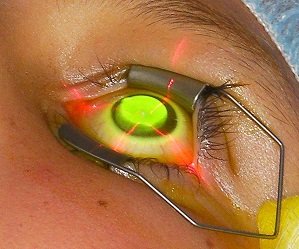
he procedure is performed in the clinic and begins with a 30-minute riboflavin loading phase. At the end, a contact lens is placed. Most patients experience mild discomfort and hazy vision for a few days. Corneal flattening occurs gradually over 6–12 months, during which some vision fluctuations are expected. Studies show vision can improve by up to two lines over time.
How do I schedule a Crosslinking procedure?
An evaluation is needed to confirm keratoconus, which involves a specialized test called Pentacam Topography. This quick exam is performed in the clinic and only takes a few minutes. Use the link below to schedule your appointment.